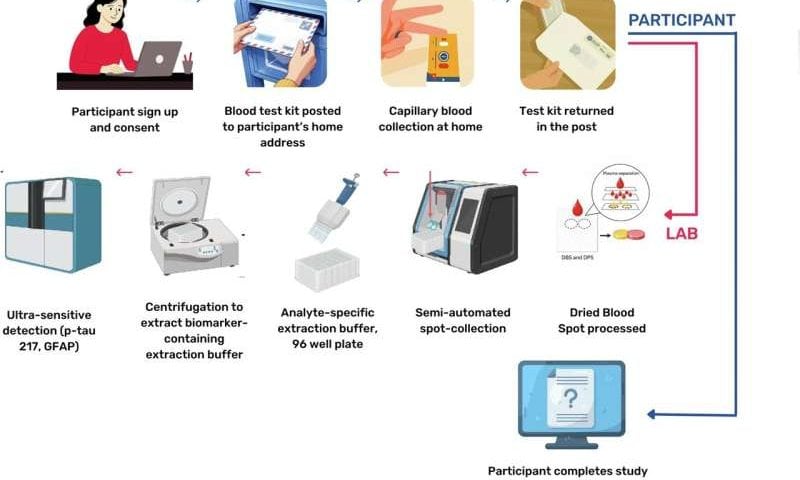
Record Breaking Clinical Milestone Achievements Transform Global Drug Development Landscape
The pharmaceutical industry is experiencing an unprecedented transformation as regulatory agencies worldwide celebrate a surge in clinical milestone achievements that are fundamentally reshaping drug development timelines and market access strategies. This remarkable shift represents more than statistical progress—it signals a new era of accelerated innovation that promises to bring life-changing treatments to patients faster than ever before.
Recent data from major regulatory bodies including the FDA, EMA, and Health Canada reveals that clinical milestone completion rates have reached historic highs, with breakthrough therapy designations and accelerated approval pathways driving this exceptional performance. The convergence of advanced clinical trial methodologies, real-world evidence integration, and enhanced regulatory science has created an environment where each clinical milestone achieved carries greater weight and predictive value for ultimate market success.
What makes this current landscape particularly compelling is the quality and diversity of programs reaching critical development phases. Oncology continues to dominate clinical milestone achievements, but rare disease treatments, gene therapies, and precision medicine approaches are increasingly contributing to this growth trajectory. The ripple effects extend beyond individual companies to influence investor confidence, partnership strategies, and global market dynamics in ways that industry veterans describe as transformational.
International harmonization efforts have played a crucial role in amplifying the impact of each clinical milestone reached by development teams. The implementation of streamlined regulatory pathways across multiple jurisdictions means that a single pivotal trial success can now trigger coordinated review processes that were previously sequential and time-consuming. This efficiency has created a multiplier effect where clinical milestone achievements generate broader market momentum and accelerated patient access timelines.
The economic implications of these clinical milestone trends cannot be overstated. Market analysts report that successful clinical milestone achievement has become the primary driver of biotechnology valuations, often resulting in immediate share price adjustments that reflect the enhanced probability of commercial success. This heightened correlation between clinical progress and market value has attracted new categories of investors who previously viewed pharmaceutical development as too unpredictable for their risk tolerance.
Technology integration has emerged as a critical factor in clinical milestone optimization, with artificial intelligence, machine learning, and advanced data analytics enabling more precise trial design and execution. These technological advances are not merely improving efficiency—they are fundamentally changing how clinical milestone endpoints are defined, measured, and validated. The result is a more robust and predictable development process that regulatory agencies increasingly trust and expedite.
International market dynamics are being reshaped by the geographic distribution of clinical milestone achievements. While traditional pharmaceutical hubs continue to generate significant clinical progress, emerging markets are increasingly contributing breakthrough developments that capture global attention. This geographic diversification is creating new partnership opportunities and challenging established market hierarchies in ways that promise long-term benefits for global patient populations.
The regulatory environment itself continues to evolve in response to clinical milestone trends, with agencies implementing new frameworks that recognize the predictive value of early-stage successes. Adaptive trial designs, master protocols, and platform studies are becoming standard approaches that enable more efficient clinical milestone progression while maintaining rigorous safety and efficacy standards. These methodological advances represent a fundamental shift toward evidence-based flexibility that benefits both developers and patients.
Looking ahead, the clinical milestone landscape appears positioned for continued growth as pipeline depth and diversity reach unprecedented levels. The combination of scientific advancement, regulatory innovation, and market dynamics has created a virtuous cycle where success breeds further success, accelerating the pace of medical breakthrough delivery to patients worldwide. This momentum represents more than industry progress—it embodies the realization of pharmaceutical development’s ultimate promise to transform human health through scientific excellence and strategic execution.